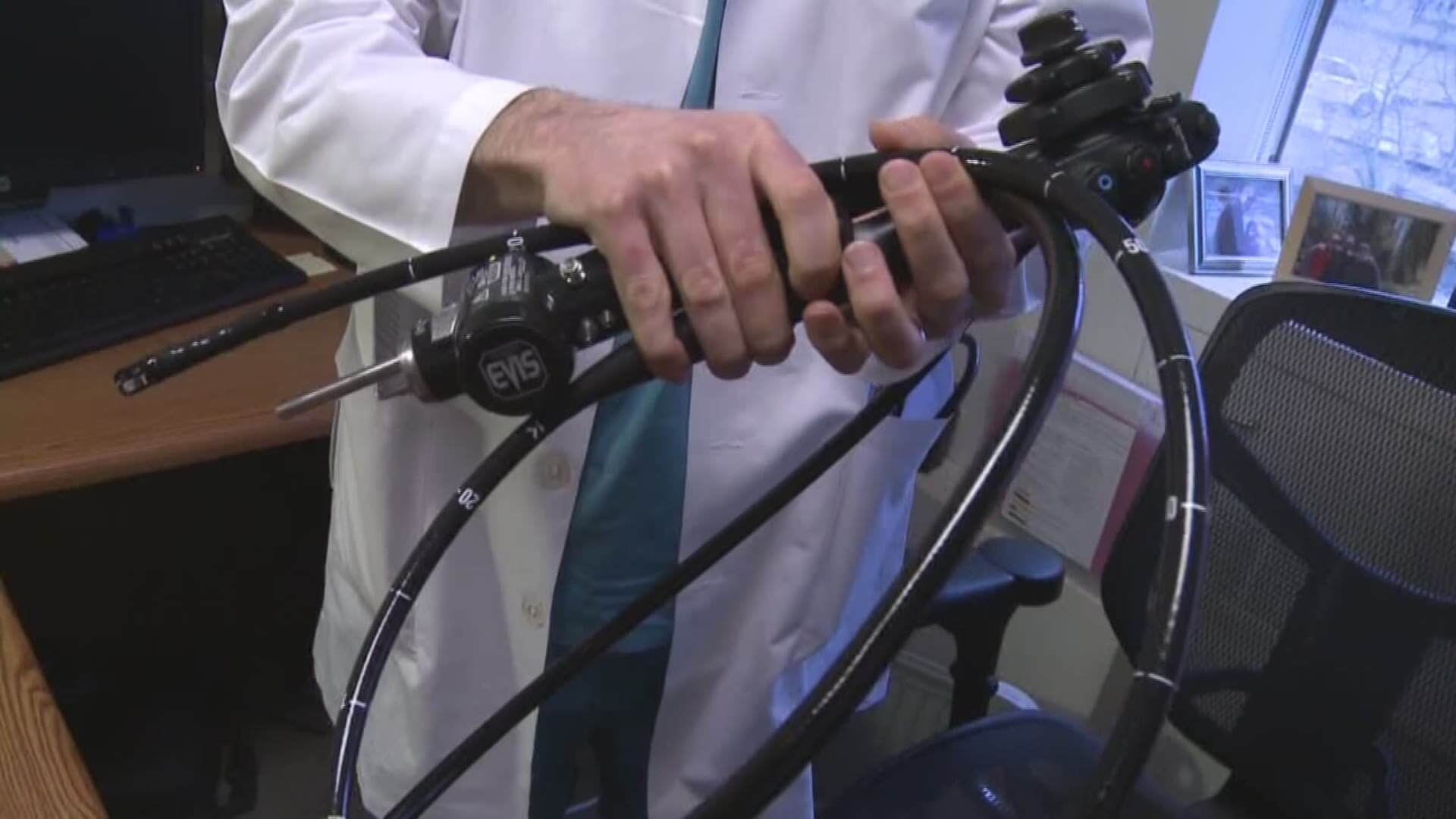
Around this time last year, the U.S. Food and Drug Administration (FDA) sent a warning to doctors and patients that a popular device called a duodenoscope was linked to a number of deadly infections. In total, the FDA reports contaminated duodenoscopes caused 35 deaths. However, a new device looks to completely eradicate deaths caused by contaminated duodenscopes.
Duodenoscopes are small scopes with cameras on the end that can be fed through the mouth and down to the intestines to give doctors a complete view of a patient’s colon. They are used in over 700,000 procedures each year and help diagnose conditions of the bile ducts and pancreatic ducts.
But duodenoscopes don’t come cheap. The devices cost approximately $40,000 each, so hospitals and healthcare facilities use them on many patients, sterilizing them between each use. The problem with duodenscopes is many are designed in a way that makes them challenging to properly sterilize, meaning patients undergo procedures with duodenscopes that are not completely sterile. Contaminants like MRSA and other superbugs can easily spread via contaminated duodenscopes and cause life-threatening injuries to patients.
But all of this is about to change. The FDA recently approved a few new, much cheaper duodenscopes – as low as $250 per device – meant to be disposable. With each patient receiving his or her own duodenscope, they won’t have to worry about what contaminants the device picked up with the previous patient.
One of the largest manufacturers of duodenscopes in the U.S., Olympus, currently faces lawsuits by patients who received infections from the device. Plaintiffs believe the company knew the device could cause deadly infections, but failed to warn patients. The company is also under investigation by federal prosecutors in New Jersey and could see criminal charges filed for fraud.