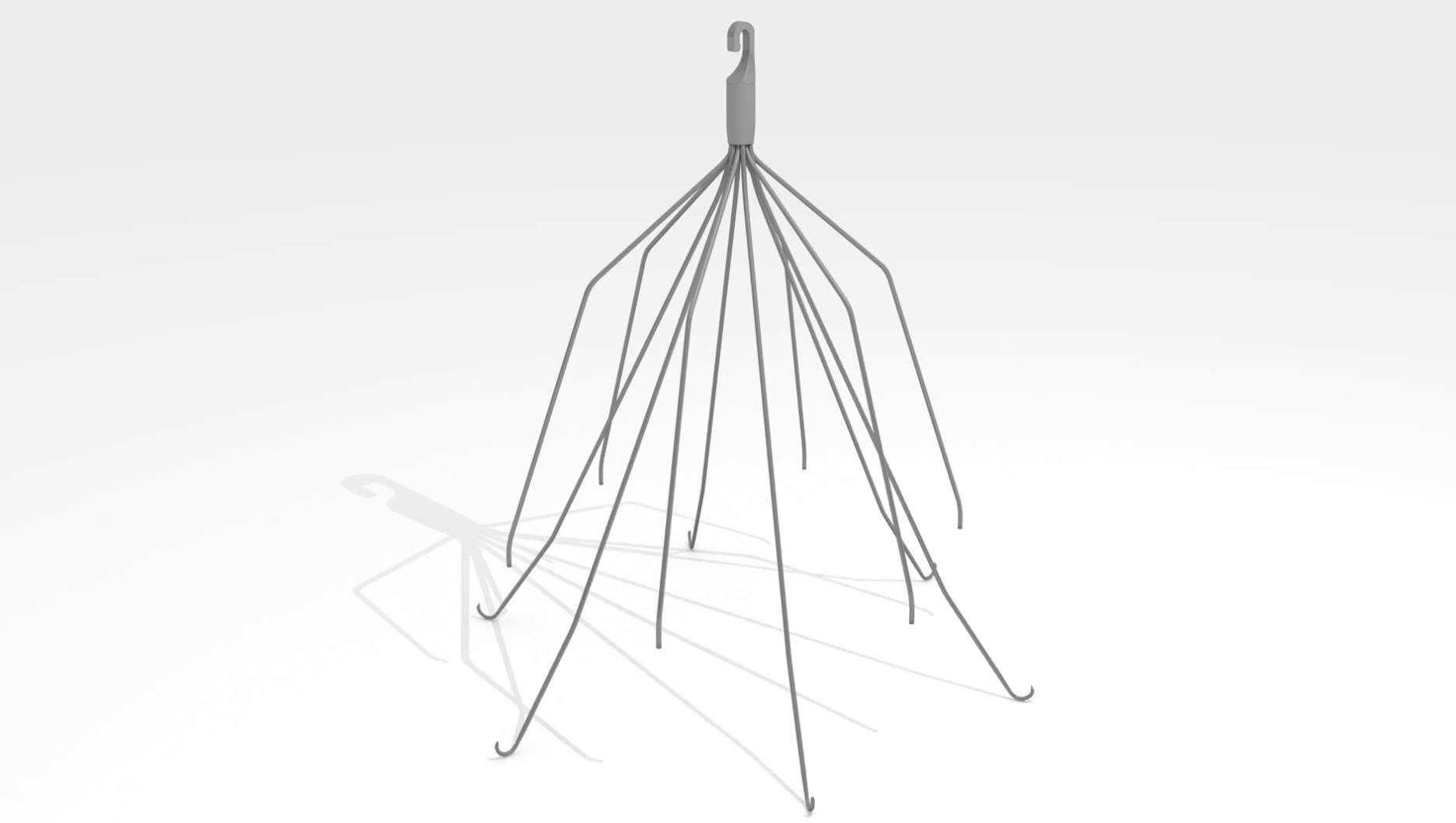
A Texas man joins hundreds of other plaintiffs in a lawsuit against Cook Medical for the company’s defective IVC filters. The man received Cook’s retrievable Günther Tulip IVC filter to reduce his risk of pulmonary embolism, but experienced complications from the device requiring further medical treatment.
Patients who experience extreme trauma or undergo surgery are at a much higher risk of developing blood clots in their lower extremities. These blood clots could travel through the body and into the heart or lungs causing pulmonary embolism (PE). For these patients, they cannot use normal methods to reduce their risk of PE like anticoagulants because they would also pose a bleeding risk. IVC filters are small cage-like devices designed to catch blood clots before they travel into the heart or lungs and cause serious complications or death while still allowing blood to flow through the device.
While IVC filters are designed to help patients who are at risk of PE and cannot take blood thinners, the devices have been known to fail inside the body. Between 2005 and 2010, the Food and Drug Administration (FDA) received 921 reports of device failure of IVC filters.
Of these reports, 328 involved device migration, 146 involved device embolization, 70 involved perforation of the inferior vena cava wall, and 56 involved filter fracture. The FDA found the longer IVC filters are in the body, the higher the chances of device failure. This led the FDA to recommend doctors remove IVC filters in patients immediately after the threat of PE had passed.
Reports of IVC filter failure continued to climb after the FDA’s safety statement and there are now hundreds of IVC filter lawsuits alleging manufacturers failed to warn doctors and patients of the risks of device failure.
The first of these manufacturers to head to court is Cook Medical. The first bellwether trials are scheduled for September, but Cook has asked for a postponement to review cases for a possible settlement conference. These first trials will likely set an important precedent for other lawsuits against C.R. Bard, Cordis Corporation, and Boston Scientific.